Can You Use Roebic Lye for Soap Making?
RusticWise is supported by its readers. When you purchase through links on our site, we may earn an affiliate commission. As an Amazon Associate, we earn from qualifying purchases. Thank You!
If you’re looking to buy lye for soap making (aka caustic soda), you might see some options at your local hardware store. Roebic lye, also known as Heavy Duty Crystal Drain Opener is labelled as 100 percent lye. It’s manufactured by Roebic Laboratories.
If you’re wondering if it’s safe to use Roebic lye for soap making, the short answer is yes. Lye (sodium hydroxide) is an essential ingredient when making homemade soap from scratch using either cold process or hot process methods. As long as you double-check the label to ensure it contains only sodium hydroxide, you’re good to go.
Let’s take a closer look at what you need to know about lye and soap making and the importance of using pure lye to create a better (and safe) bar of soap.
What exactly is Roebic lye used for?
Roebic lye, also known as Roebic Professional Strength Crystal Drain Opener, or Roebic Crystal Drain Opener, is an industrial-strength drain cleaner marketed for commercial use.¹
Labelled as 100 percent lye (also known as sodium hydroxide, caustic soda, or NaOH), this product eats away at clogged drains and banishes grease.
If you’re familiar with lye, you know that this stuff is powerful! If you’re looking for an effective solution for drain cleaning, this is the product to use. Use with caution as it can eat away at clothing, cause skin burns or irritation, and harm your eyes.
Roebic Laboratories also manufactures several other products, including treatment for wastewater systems (such as septic tanks and cesspools), products for cleaning RVs, and root killers. They also developed products with unique bacterial cultures designed to eat away at fat, oil, and grease—the primary causes of clogged drains.
Note: While Roebic Heavy Duty Crystal Drain Opener is marketed as a drain cleaner, does this mean you can use any type of drain cleaner to make soap? No. Most other drain cleaners at your local hardware store likely list sodium hydroxide as one ingredient, but they often contain a chemical cocktail that’s NOT safe for soap making. (After all, nobody wants to rub caustic chemicals onto their skin, right?)
You might like
Roebic Lye Professional Strength Crystal Drain Opener
- Contains 100% sodium hydroxide (caustic soda). Not for use in toilets, garbage disposals or with aluminum, lead, zinc or galvanized pipes.
- Melts and destroys grease, other kitchen clogs and will keep drains clear.
- 2-lbs.
Found on Amazon
Check Current Price
Those in Canada and the UK should be taken to the product listing in your region.
Lye safety precautions
If you’re a new soap maker, make sure you read up on lye safety first as it’s a highly corrosive compound that can harm your skin, eyes, and cause severe burns if not used safely.
Here are a few things to keep in mind:
- Avoid using any aluminum pots, bowls, or utensils when making soap. Lye reacts strongly with aluminum.
- Always wear protective equipment when handling lye. This includes gloves, a long-sleeved shirt, and safety goggles.
- Work in a well-ventilated room.
- Always add lye to water when making your lye solution (and not the other way around).
- Store lye in a dry, cool place in an airtight and clearly labelled container. Ensure it’s in a safe location that’s out of reach of children and pets.

What’s in Roebic lye: a closer look
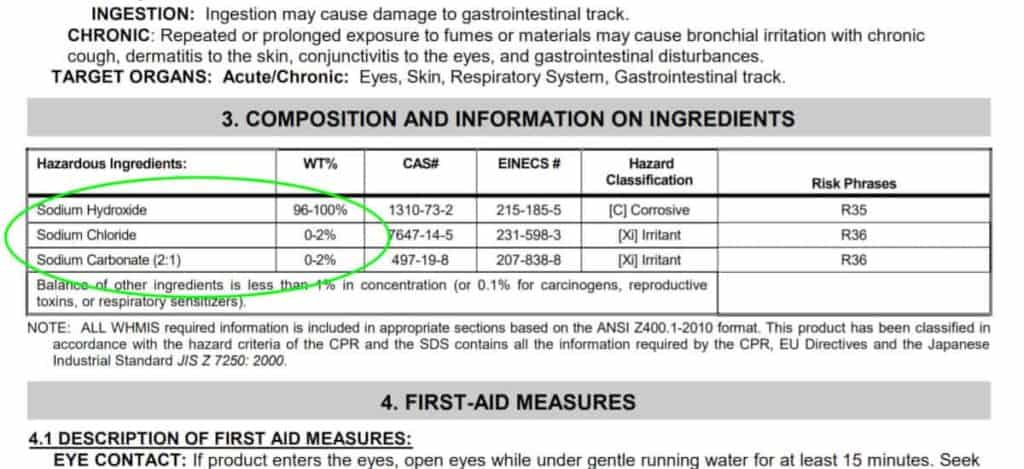
Curious about what’s really in Roebic lye, I took a closer look at the material safety data sheet (MSDS).²
As you can see, it contains 96–100 percent sodium hydroxide (lye). It also contains between 0 and 2 percent sodium chloride; and between 0 and 2 percent sodium carbonate.
Don’t let this alarm you, however.
Sodium chloride (NaCl) is essentially table salt.
Sodium carbonate (Na₂CO₃) is also called soda ash or washing soda. It’s a naturally occurring alkaline salt, or a salt of carbonic acid. According to PubChem, “Sodium carbonate is an organic sodium salt and a carbonate salt.” When sodium carbonate is dissolved in water, it makes sodium hydroxide and carbonic acid.³
Essential Depot, a popular online supplier of soap making supplies and lye, carries lye products with a minimum strength of 96 percent.⁴
When I compare the ingredient breakdown of Roebic Crysyal Drain Opener to the MSDS sheet of a food grade lye from Essential Depot, it shows the same breakdown of the composition of ingredients:
- 96 to 100 percent sodium hydroxide
- 0 to 2 percent sodium chloride
- 0 to 2 percent sodium carbonate
Note: While many labels of both technical-grade and food-grade sodium hydroxide state they are 100 percent sodium hydroxide, most MSDS’s will allow for a discrepancy for trace oxides (sodium chloride and sodium carbonate).
What kind of lye do you need for soap making?
In a nutshell, you need pure sodium hydroxide to make a batch of handmade soap. The purer, the better some would argue.
There’s much debate in the soap making community about whether you should use a technical-grade product like Roebic lye vs. a food grade lye.
The difference is in the purity level of the lye. Food-grade lye (which is used to process foods such as pretzels and bagels) are held to a higher standard as it comes in contact with food.
According to Essential Depot, a food-grade lye, “has a lower parts per million of heavy metal impurities.”⁴
Most food-grade lyes are about 96 to 99 percent pure. Technical-grade lyes may carry slightly more impurities.
Do you need to use food-grade lye for making homemade soap?
No, you aren’t required to use food-grade lye to make a batch of homemade soap. Food-grade lye costs slightly more than technical-grade lye. However, many people prefer it from a soap maker’s perspective—it provides extra peace of mind, and some argue that it makes for a better bar of soap.
If you’re stocking up on craft supplies from an online retailer, you can pick up a bottle of lye flakes, along with quality coconut oil, olive, oil, palm oil, and any essential oils you need to make handmade soap.
On the flip side, many people say there really is little to no difference in the finished product if you’re using food grade or technical grade lye.
To be honest, I would say to use whatever is available to you (so long as it contains only pure lye).
You might like
The Boyer Corporation Red Crown Lye 2 Lbs (Pack of 2)
- 2 pack of Red Crown 2 lbs lye – a total of 4 lbs.
- High test lye for making handcrafted soaps.
- Made in the USA.
Found on Amazon
Check Current Price
Those in Canada and the UK should be taken to the product listing in your region.
Why you might not want to use Roebic lye for making soap
In essence, Roebic Crystal drain cleaner is not cheap. For those of you who live in rural areas and don’t have access to many local stores that sell lye (or don’t want to pay for the shipping costs of buying online), this may be one of your only options if you want to make a batch of homemade soap.
Otherwise, sourcing lye in bulk from a soap making supplier, online retailer, or a local supplier (if you have one) is the best, most cost-effective way to buy lye. (Of course, buying bulk lye only makes sense if you’re planning on making many batches of soap.)
Another reason you may not want to use Roebic lye is from a soap purist’s point of view. If you prefer to use only food-grade lye in your homemade batch, that’s a personal preference.
Tip: Lye is not the only ingredient that can add impurities to your soap recipe. The quality of water you use plays a large role, too. Stick with using distilled water for making bar soap.
Is lye soap good for your skin?
It sounds crazy that a caustic chemical like lye is a key ingredient in soap, a product that we rub on our faces and body.
Through a wonderful scientific transformation known as the saponification process, lye when combined with water and fats, makes soap. The lye is the magic ingredient that binds water and fats/oils together. Without lye, you would just have a gooey mess.
Once your batch of soap has completely saponified (after you’ve carefully followed all instructions, measured your ingredients, and allowed the soap to cure), you’ll have bars of wonderful handmade soap that are safe to use.
The caustic soda will be completely neutralized.
Where do you buy lye for soap making?
You can find food-grade lyes in some local craft and baking stores (such as Michaels), and they’re readily available online through soap suppliers such as Essential Depot and Wholesale Supplies Plus.
You’ll find technical-grade lyes in most hardware stores, soap making stores, and also online.
And of course, you can find lye for soap making on Amazon.
👉Check out our article on Where to Buy Sodium Hydroxide for Soapmaking.
If you don’t want to handle lye when making soap . . .
If the thought of handling a caustic substance like lye freaks you out, try making melt and pour soap. This is a great way for beginner soap makers to make handcrafted suds using fully saponified soap bases (the lye has already been turned to soap!).
All you need to do is melt your favorite soap bases, add any essential oils, additives, or colorants, and pour into molds.
Related questions
Can you use sodium carbonate instead of sodium hydroxide for soap?
No, you can’t use sodium carbonate (also known as soda ash) in place of sodium hydroxide (lye/caustic soda). While the two chemicals have similarities (both are caustic alkalis), sodium hydroxide is much more alkaline.
Sodium hydroxide has an approximate pH between 13 and 14, while sodium carbonate has a pH of 11–12. On the pH scale, each unit increase represents a change in hydrogen atoms by a factor of 10. A two unit increase would mean a change in hydrogen atoms by a factor of 100⁵.
You could not safely or reliably substitute the two ingredients in a soap recipe without having disastrous results.
What’s the difference between sodium hydroxide and potassium hydroxide?
Confusingly, both potassium hydroxide (KOH) and sodium hydroxide (NaOH) are referred to as lye. While both are used to make soap, potassium hydroxide is commonly used to make liquid soap or soft soaps. If you’re looking to make homemade cold process soap (or hot process), you’ll be using sodium hydroxide lye.
Read more about the differences between sodium hydroxide and potassium hydroxide.
What can replace lye in soap?
Sadly, there is no true replacement for sodium hydroxide (or potassium hydroxide) in a soap recipe. Lye is an integral part of the handcrafted soap making process to bind oils/fats with water to create soap molecules.
If you don’t want to handle lye at all, try making melt and pour soap. While lye is an ingredient used to make the premade soap bases, you don’t have to handle any lye yourself at home with the M & P process.
New to making soap? 🧼❓
👉We have a fantastic overview on the whole soapmaking process here: read our Timeless Guide To Soapmaking.
If you would like to see our soapmaking posts organized by topic type, see our Soapmaking Collection.
Would you like more timeless tips via email?
Fun tips to help you live an independent, self-sustaining lifestyle. Opt-out at any time.


References
- Roebic Laboratories, About Us, https://www.roebic.com/about-us.shtml. Accessed December 2022.
- Roebic Laboratories, Professional Strength Crystal Drain Opener, Material Safety Data Sheet, https://www.roebic.com/heavy-duty-crystal-drain-opener.shtml. Accessed December 2022.
- PubChem (National Library of Medicine), Sodium carbonate, https://pubchem.ncbi.nlm.nih.gov/compound/Sodium-carbonate. Accessed December 2022.
- Essential Depot, Lye NaOH – KOH, https://www.essentialdepot.com/category/sodium-hydroxide-components.html?. Accessed December 2022.
- Lumen, The pH Scale, https://courses.lumenlearning.com/wmopen-nmbiology1/chapter/the-ph-scale/. Accessed December 2022.

Author: Theresa Tesolin
Theresa is co-founder of RusticWise. She helps people unleash their inner DIY spirit by encouraging them to get dirty and make or grow something from scratch.